| کد مقاله | کد نشریه | سال انتشار | مقاله انگلیسی | نسخه تمام متن |
|---|---|---|---|---|
| 1361349 | 981461 | 2008 | 8 صفحه PDF | دانلود رایگان |
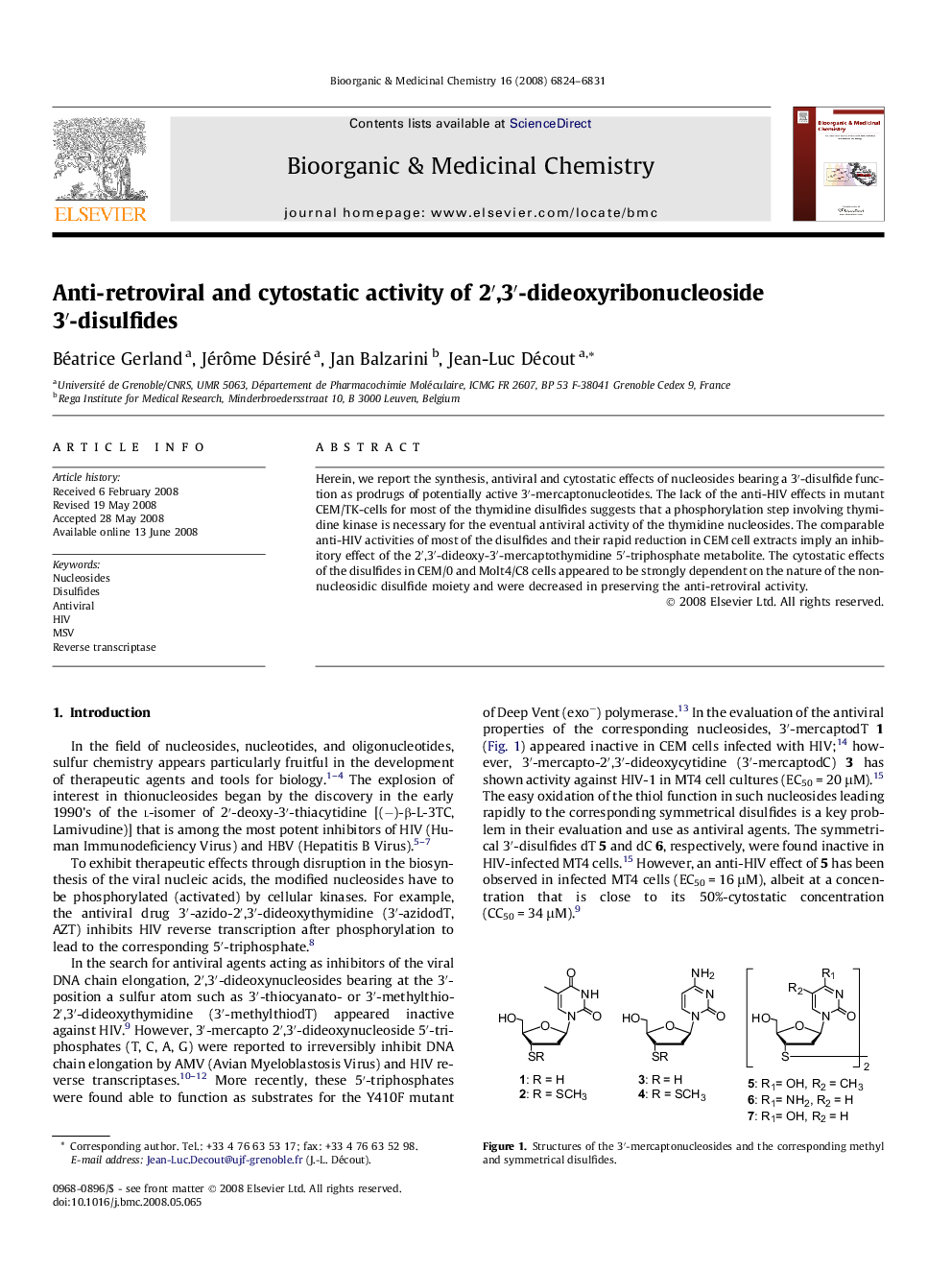
Herein, we report the synthesis, antiviral and cytostatic effects of nucleosides bearing a 3′-disulfide function as prodrugs of potentially active 3′-mercaptonucleotides. The lack of the anti-HIV effects in mutant CEM/TK-cells for most of the thymidine disulfides suggests that a phosphorylation step involving thymidine kinase is necessary for the eventual antiviral activity of the thymidine nucleosides. The comparable anti-HIV activities of most of the disulfides and their rapid reduction in CEM cell extracts imply an inhibitory effect of the 2′,3′-dideoxy-3′-mercaptothymidine 5′-triphosphate metabolite. The cytostatic effects of the disulfides in CEM/0 and Molt4/C8 cells appeared to be strongly dependent on the nature of the non-nucleosidic disulfide moiety and were decreased in preserving the anti-retroviral activity.
Figure optionsDownload as PowerPoint slide
Journal: Bioorganic & Medicinal Chemistry - Volume 16, Issue 14, 15 July 2008, Pages 6824–6831