| کد مقاله | کد نشریه | سال انتشار | مقاله انگلیسی | نسخه تمام متن |
|---|---|---|---|---|
| 1408147 | 1501723 | 2015 | 7 صفحه PDF | دانلود رایگان |
• Borate glasses are characterized by thermal, structural and optical properties.
• Raman measurement shows the functional groups of borate ions.
• Borate glasses were exhibited a strong visible emission in blue and yellow region while a weak emission was observed in red region. These emission lines are characteristics of Dy3+ ion.
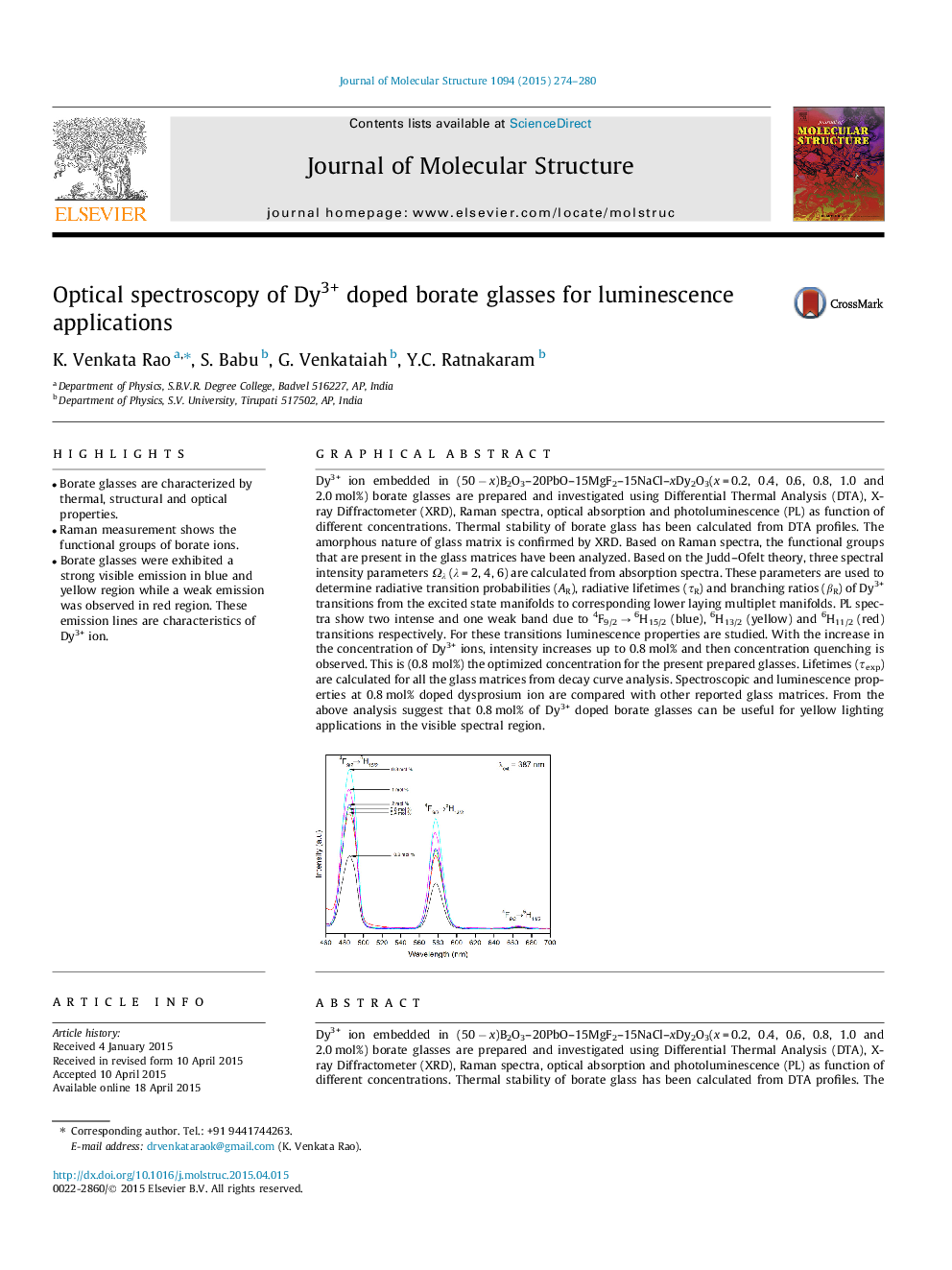
Dy3+ ion embedded in (50 − x)B2O3–20PbO–15MgF2–15NaCl–xDy2O3(x = 0.2, 0.4, 0.6, 0.8, 1.0 and 2.0 mol%) borate glasses are prepared and investigated using Differential Thermal Analysis (DTA), X-ray Diffractometer (XRD), Raman spectra, optical absorption and photoluminescence (PL) as function of different concentrations. Thermal stability of borate glass has been calculated from DTA profiles. The amorphous nature of glass matrix is confirmed by XRD. Based on Raman spectra, the functional groups that are present in the glass matrices have been analyzed. Based on the Judd–Ofelt theory, three spectral intensity parameters Ωλ (λ = 2, 4, 6) are calculated from absorption spectra. These parameters are used to determine radiative transition probabilities (AR), radiative lifetimes (τR) and branching ratios (βR) of Dy3+ transitions from the excited state manifolds to corresponding lower laying multiplet manifolds. PL spectra show two intense and one weak band due to 4F9/2 → 6H15/2 (blue), 6H13/2 (yellow) and 6H11/2 (red) transitions respectively. For these transitions luminescence properties are studied. With the increase in the concentration of Dy3+ ions, intensity increases up to 0.8 mol% and then concentration quenching is observed. This is (0.8 mol%) the optimized concentration for the present prepared glasses. Lifetimes (τexp) are calculated for all the glass matrices from decay curve analysis. Spectroscopic and luminescence properties at 0.8 mol% doped dysprosium ion are compared with other reported glass matrices. From the above analysis suggest that 0.8 mol% of Dy3+ doped borate glasses can be useful for yellow lighting applications in the visible spectral region.
Dy3+ ion embedded in (50 − x)B2O3–20PbO–15MgF2–15NaCl–xDy2O3(x = 0.2, 0.4, 0.6, 0.8, 1.0 and 2.0 mol%) borate glasses are prepared and investigated using Differential Thermal Analysis (DTA), X-ray Diffractometer (XRD), Raman spectra, optical absorption and photoluminescence (PL) as function of different concentrations. Thermal stability of borate glass has been calculated from DTA profiles. The amorphous nature of glass matrix is confirmed by XRD. Based on Raman spectra, the functional groups that are present in the glass matrices have been analyzed. Based on the Judd–Ofelt theory, three spectral intensity parameters Ωλ (λ = 2, 4, 6) are calculated from absorption spectra. These parameters are used to determine radiative transition probabilities (AR), radiative lifetimes (τR) and branching ratios (βR) of Dy3+ transitions from the excited state manifolds to corresponding lower laying multiplet manifolds. PL spectra show two intense and one weak band due to 4F9/2 → 6H15/2 (blue), 6H13/2 (yellow) and 6H11/2 (red) transitions respectively. For these transitions luminescence properties are studied. With the increase in the concentration of Dy3+ ions, intensity increases up to 0.8 mol% and then concentration quenching is observed. This is (0.8 mol%) the optimized concentration for the present prepared glasses. Lifetimes (τexp) are calculated for all the glass matrices from decay curve analysis. Spectroscopic and luminescence properties at 0.8 mol% doped dysprosium ion are compared with other reported glass matrices. From the above analysis suggest that 0.8 mol% of Dy3+ doped borate glasses can be useful for yellow lighting applications in the visible spectral region.Figure optionsDownload as PowerPoint slide
Journal: Journal of Molecular Structure - Volume 1094, 15 August 2015, Pages 274–280