| کد مقاله | کد نشریه | سال انتشار | مقاله انگلیسی | نسخه تمام متن |
|---|---|---|---|---|
| 1409280 | 1501746 | 2014 | 5 صفحه PDF | دانلود رایگان |
• The flexible ligands and metal ions play a significant role in the formation and structures of coordination compounds.
• The bpp ligand adopts a chelating-bidentate coordination mode.
• The complexes have been characterized by X-ray crystallography, elemental and thermal analyses and infrared spectroscopy.
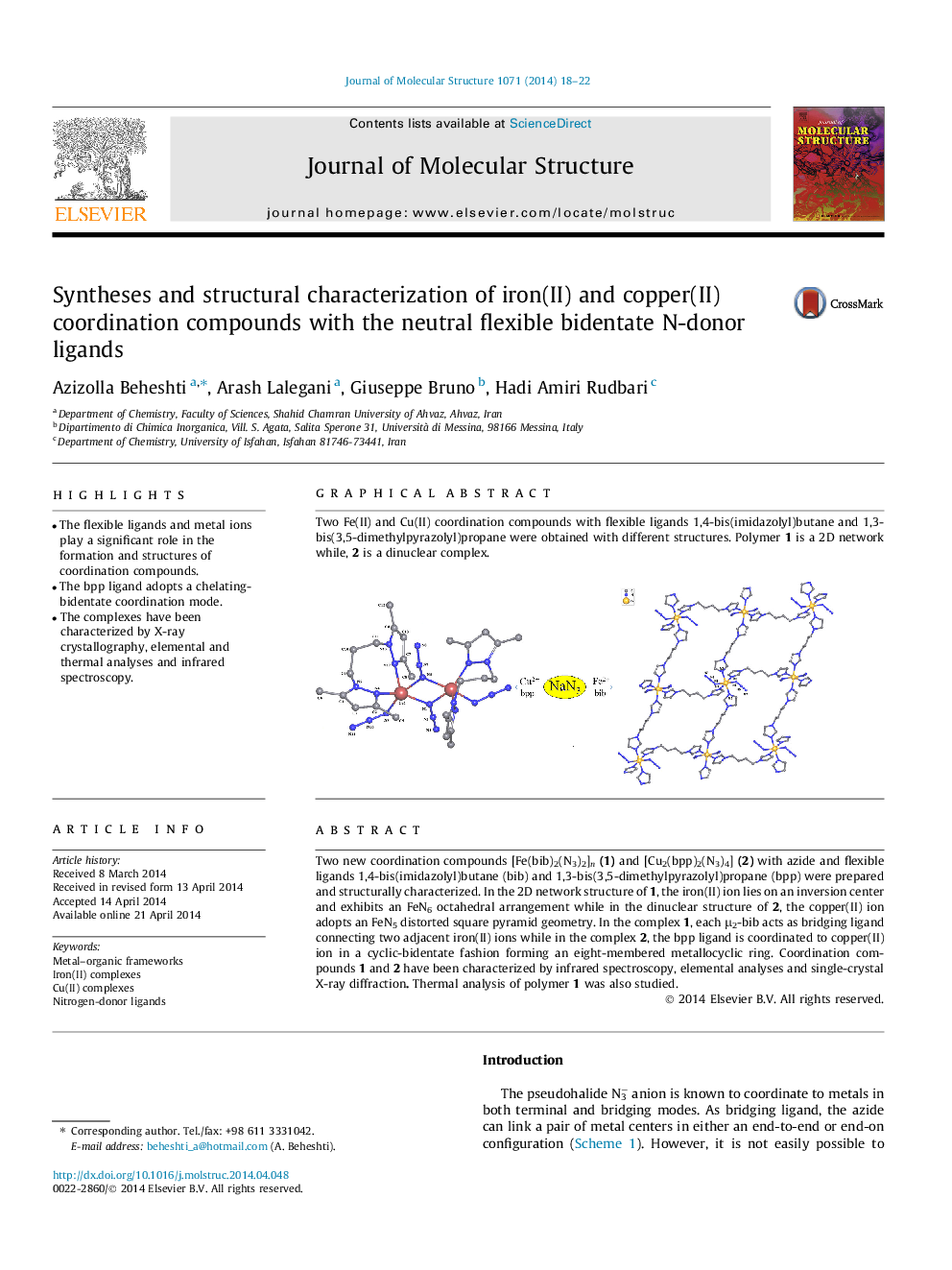
Two new coordination compounds [Fe(bib)2(N3)2]n(1) and [Cu2(bpp)2(N3)4] (2) with azide and flexible ligands 1,4-bis(imidazolyl)butane (bib) and 1,3-bis(3,5-dimethylpyrazolyl)propane (bpp) were prepared and structurally characterized. In the 2D network structure of 1, the iron(II) ion lies on an inversion center and exhibits an FeN6 octahedral arrangement while in the dinuclear structure of 2, the copper(II) ion adopts an FeN5 distorted square pyramid geometry. In the complex 1, each μ2-bib acts as bridging ligand connecting two adjacent iron(II) ions while in the complex 2, the bpp ligand is coordinated to copper(II) ion in a cyclic-bidentate fashion forming an eight-membered metallocyclic ring. Coordination compounds 1 and 2 have been characterized by infrared spectroscopy, elemental analyses and single-crystal X-ray diffraction. Thermal analysis of polymer 1 was also studied.
Two Fe(II) and Cu(II) coordination compounds with flexible ligands 1,4-bis(imidazolyl)butane and 1,3-bis(3,5-dimethylpyrazolyl)propane were obtained with different structures. Polymer 1 is a 2D network while, 2 is a dinuclear complex.Figure optionsDownload as PowerPoint slide
Journal: Journal of Molecular Structure - Volume 1071, 5 August 2014, Pages 18–22