| کد مقاله | کد نشریه | سال انتشار | مقاله انگلیسی | نسخه تمام متن |
|---|---|---|---|---|
| 1409336 | 1501751 | 2014 | 4 صفحه PDF | دانلود رایگان |
• Stability constant of the antamanide–Ba2+ cationic complex in water-saturated nitrobenzene was determined.
• Quantum mechanical DFT calculations were carried out.
• Structure of the resulting cationic complex species was predicted.
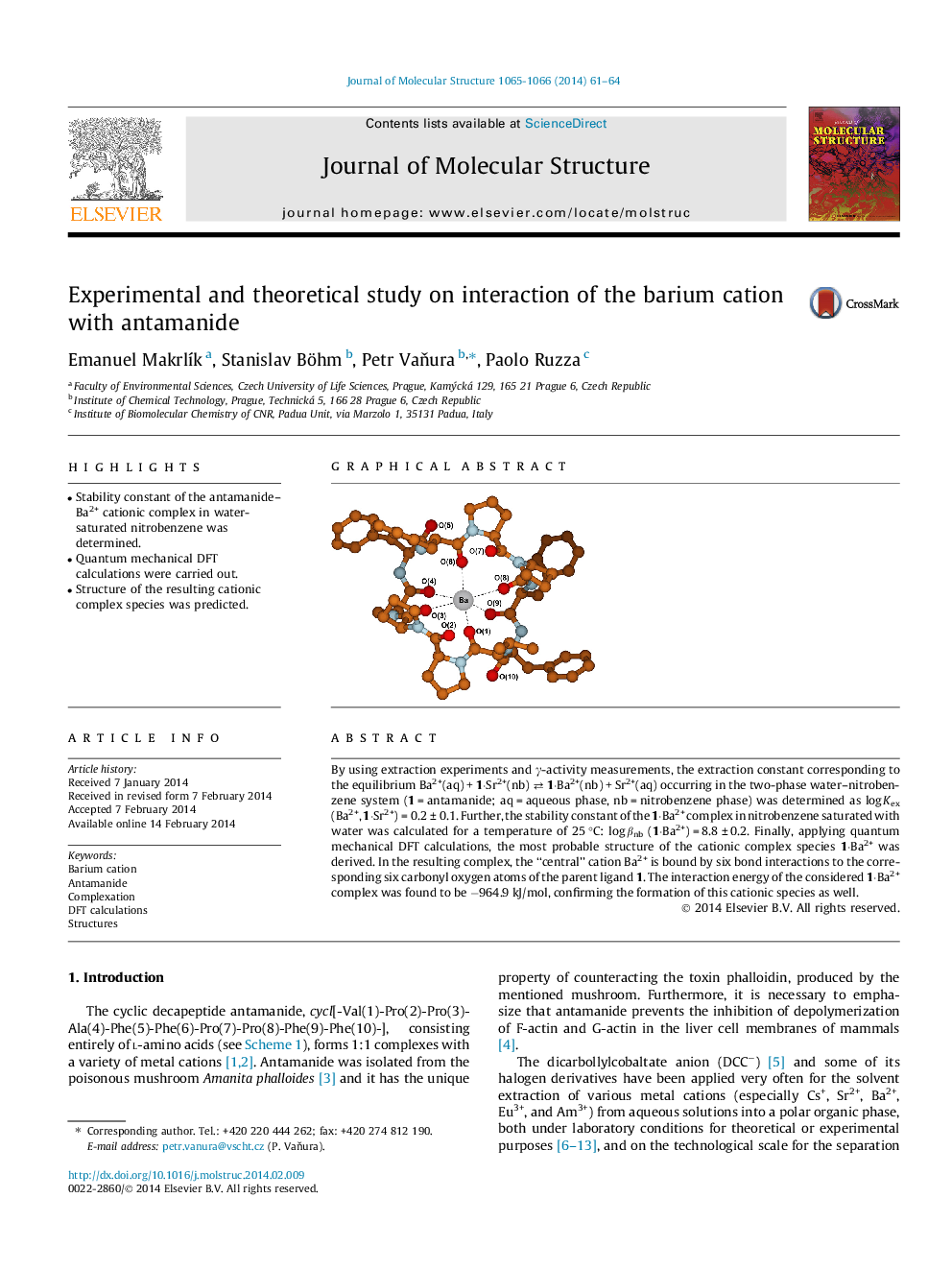
By using extraction experiments and γ-activity measurements, the extraction constant corresponding to the equilibrium Ba2+(aq) + 1⋅Sr2+(nb) ⇄ 1⋅Ba2+(nb) + Sr2+(aq) occurring in the two-phase water–nitrobenzene system (1 = antamanide; aq = aqueous phase, nb = nitrobenzene phase) was determined as log Kex (Ba2+, 1⋅Sr2+) = 0.2 ± 0.1. Further, the stability constant of the 1⋅Ba2+ complex in nitrobenzene saturated with water was calculated for a temperature of 25 °C: log βnb (1⋅Ba2+) = 8.8 ± 0.2. Finally, applying quantum mechanical DFT calculations, the most probable structure of the cationic complex species 1⋅Ba2+ was derived. In the resulting complex, the “central” cation Ba2+ is bound by six bond interactions to the corresponding six carbonyl oxygen atoms of the parent ligand 1. The interaction energy of the considered 1⋅Ba2+ complex was found to be −964.9 kJ/mol, confirming the formation of this cationic species as well.
Figure optionsDownload as PowerPoint slide
Journal: Journal of Molecular Structure - Volumes 1065–1066, 22 May 2014, Pages 61–64