| کد مقاله | کد نشریه | سال انتشار | مقاله انگلیسی | نسخه تمام متن |
|---|---|---|---|---|
| 148913 | 456424 | 2013 | 8 صفحه PDF | دانلود رایگان |
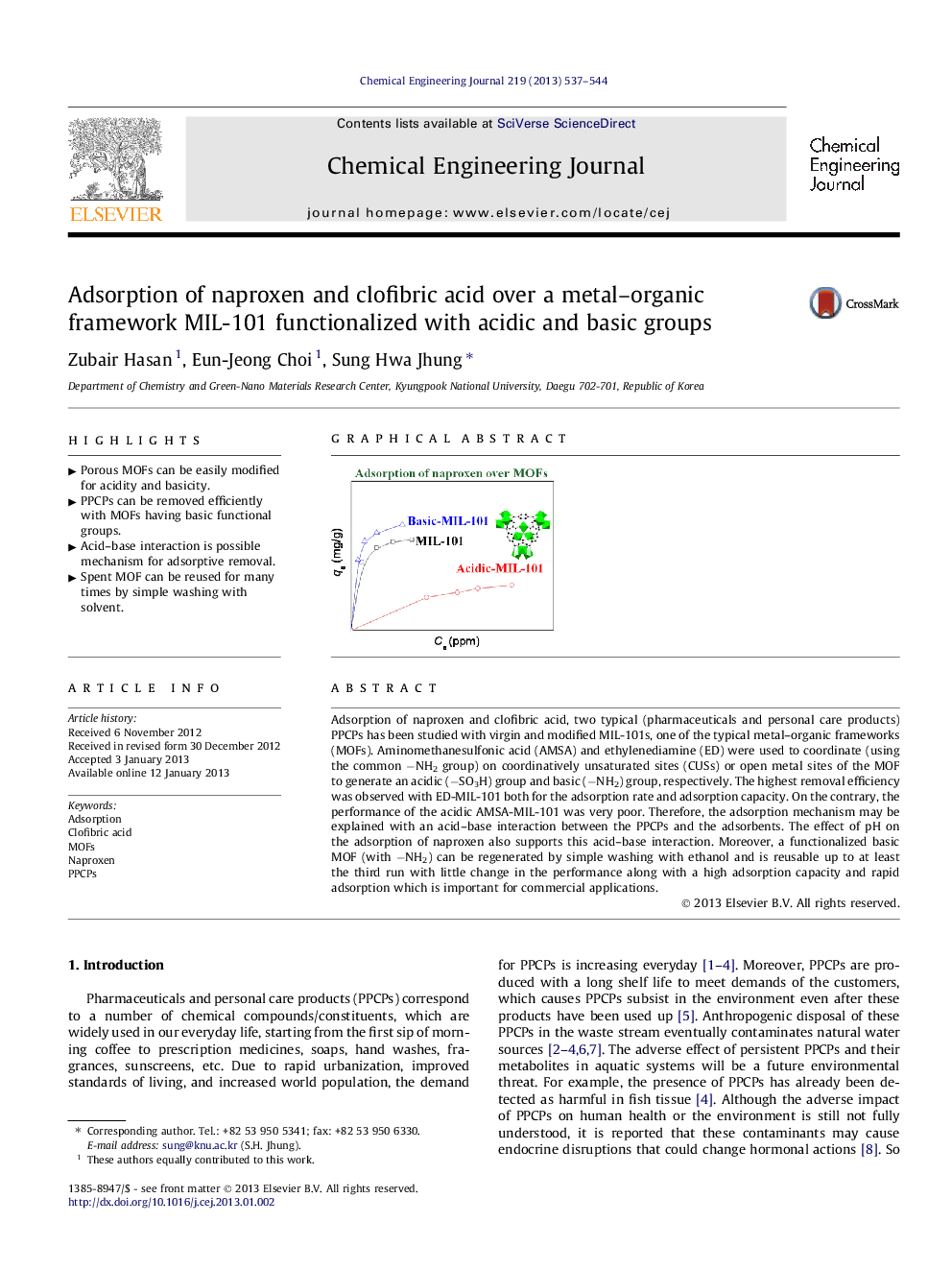
Adsorption of naproxen and clofibric acid, two typical (pharmaceuticals and personal care products) PPCPs has been studied with virgin and modified MIL-101s, one of the typical metal–organic frameworks (MOFs). Aminomethanesulfonic acid (AMSA) and ethylenediamine (ED) were used to coordinate (using the common −NH2 group) on coordinatively unsaturated sites (CUSs) or open metal sites of the MOF to generate an acidic (−SO3H) group and basic (−NH2) group, respectively. The highest removal efficiency was observed with ED-MIL-101 both for the adsorption rate and adsorption capacity. On the contrary, the performance of the acidic AMSA-MIL-101 was very poor. Therefore, the adsorption mechanism may be explained with an acid–base interaction between the PPCPs and the adsorbents. The effect of pH on the adsorption of naproxen also supports this acid–base interaction. Moreover, a functionalized basic MOF (with −NH2) can be regenerated by simple washing with ethanol and is reusable up to at least the third run with little change in the performance along with a high adsorption capacity and rapid adsorption which is important for commercial applications.
Figure optionsDownload as PowerPoint slideHighlights
► Porous MOFs can be easily modified for acidity and basicity.
► PPCPs can be removed efficiently with MOFs having basic functional groups.
► Acid–base interaction is possible mechanism for adsorptive removal.
► Spent MOF can be reused for many times by simple washing with solvent.
Journal: Chemical Engineering Journal - Volume 219, 1 March 2013, Pages 537–544