| کد مقاله | کد نشریه | سال انتشار | مقاله انگلیسی | نسخه تمام متن |
|---|---|---|---|---|
| 5371179 | 1503940 | 2012 | 8 صفحه PDF | دانلود رایگان |
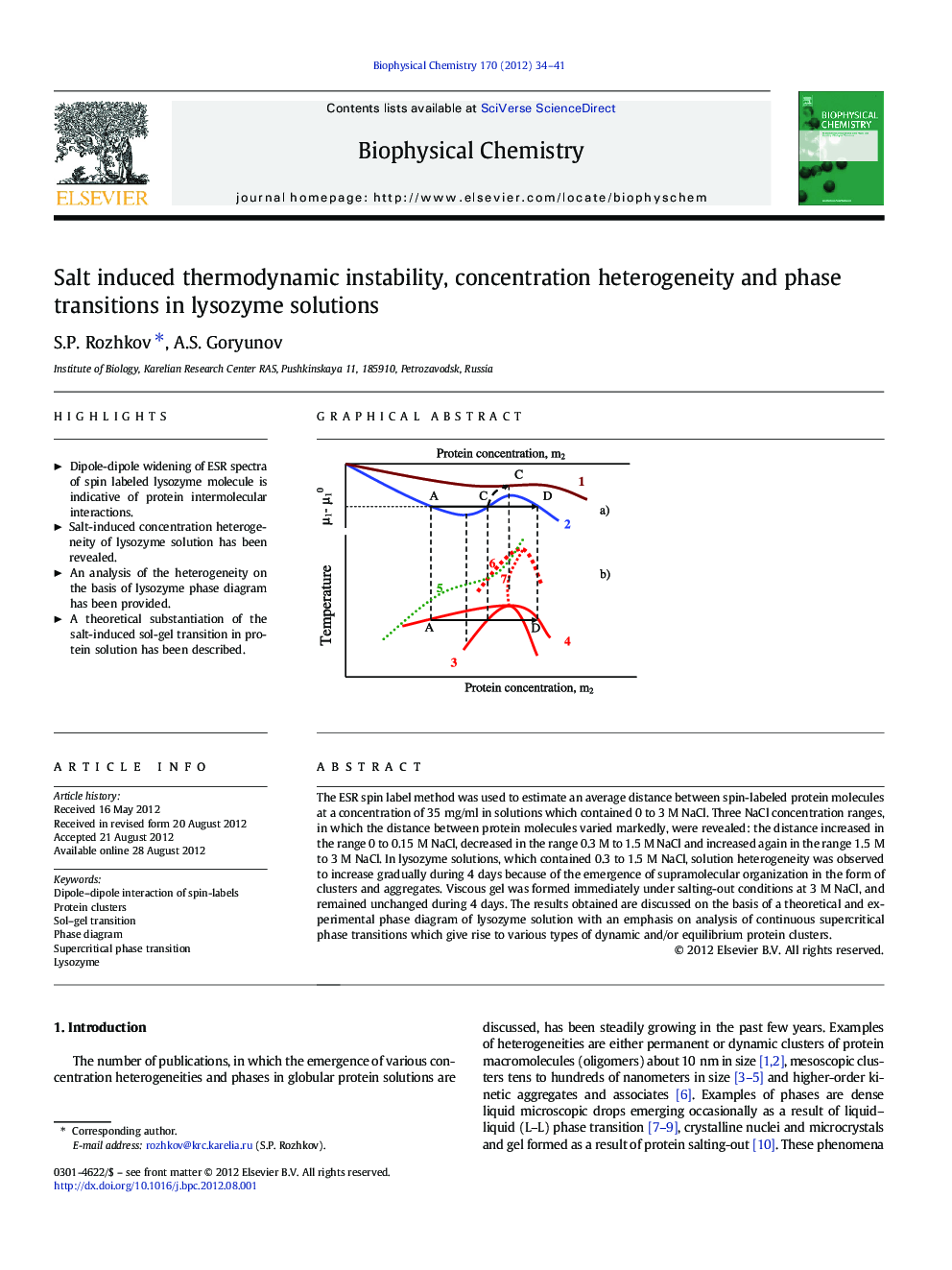
The ESR spin label method was used to estimate an average distance between spin-labeled protein molecules at a concentration of 35Â mg/ml in solutions which contained 0 to 3Â M NaCl. Three NaCl concentration ranges, in which the distance between protein molecules varied markedly, were revealed: the distance increased in the range 0 to 0.15Â Ð NaCl, decreased in the range 0.3 Ð to 1.5 Ð NaCl and increased again in the range 1.5Â Ð to 3Â Ð NaCl. In lysozyme solutions, which contained 0.3 to 1.5Â Ð NaCl, solution heterogeneity was observed to increase gradually during 4Â days because of the emergence of supramolecular organization in the form of clusters and aggregates. Viscous gel was formed immediately under salting-out conditions at 3Â Ð NaCl, and remained unchanged during 4Â days. The results obtained are discussed on the basis of a theoretical and experimental phase diagram of lysozyme solution with an emphasis on analysis of continuous supercritical phase transitions which give rise to various types of dynamic and/or equilibrium protein clusters.
Highlights⺠Dipole-dipole widening of ESR spectra of spin labeled lysozyme molecule is indicative of protein intermolecular interactions. ⺠Salt-induced concentration heterogeneity of lysozyme solution has been revealed. ⺠An analysis of the heterogeneity on the basis of lysozyme phase diagram has been provided. ⺠A theoretical substantiation of the salt-induced sol-gel transition in protein solution has been described.
Journal: Biophysical Chemistry - Volume 170, AugustâSeptember 2012, Pages 34-41