| کد مقاله | کد نشریه | سال انتشار | مقاله انگلیسی | نسخه تمام متن |
|---|---|---|---|---|
| 593785 | 1453954 | 2013 | 7 صفحه PDF | دانلود رایگان |
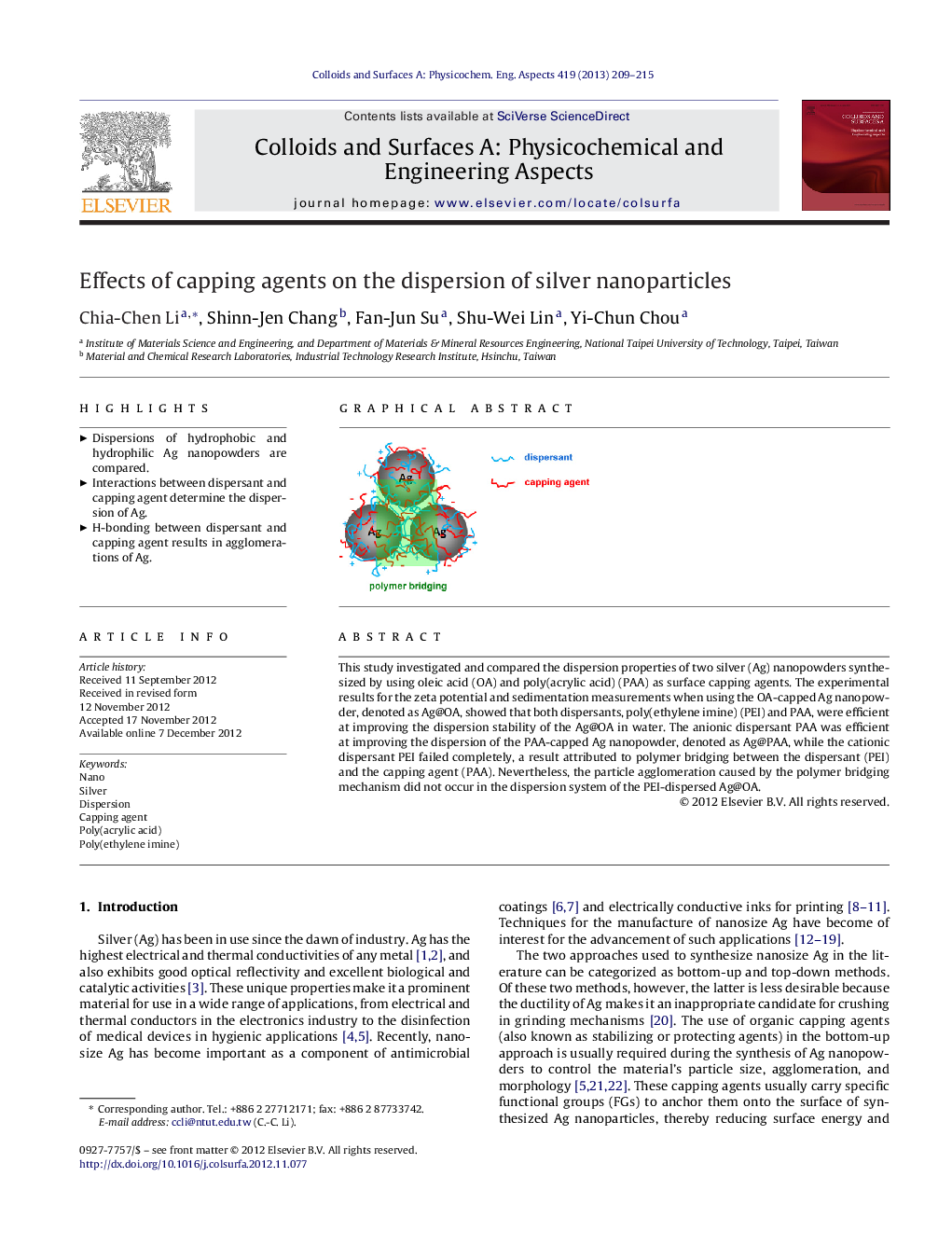
This study investigated and compared the dispersion properties of two silver (Ag) nanopowders synthesized by using oleic acid (OA) and poly(acrylic acid) (PAA) as surface capping agents. The experimental results for the zeta potential and sedimentation measurements when using the OA-capped Ag nanopowder, denoted as Ag@OA, showed that both dispersants, poly(ethylene imine) (PEI) and PAA, were efficient at improving the dispersion stability of the Ag@OA in water. The anionic dispersant PAA was efficient at improving the dispersion of the PAA-capped Ag nanopowder, denoted as Ag@PAA, while the cationic dispersant PEI failed completely, a result attributed to polymer bridging between the dispersant (PEI) and the capping agent (PAA). Nevertheless, the particle agglomeration caused by the polymer bridging mechanism did not occur in the dispersion system of the PEI-dispersed Ag@OA.
Capping agent interacts with the latter-added dispersant, resulting in a heavy agglomeration of silver nanoparticles.Figure optionsDownload as PowerPoint slideHighlights
► Dispersions of hydrophobic and hydrophilic Ag nanopowders are compared.
► Interactions between dispersant and capping agent determine the dispersion of Ag.
► H-bonding between dispersant and capping agent results in agglomerations of Ag.
Journal: Colloids and Surfaces A: Physicochemical and Engineering Aspects - Volume 419, 20 February 2013, Pages 209–215