| کد مقاله | کد نشریه | سال انتشار | مقاله انگلیسی | نسخه تمام متن |
|---|---|---|---|---|
| 1165112 | 1491017 | 2014 | 6 صفحه PDF | دانلود رایگان |
• We firstly inducted the hydrolysis reaction between SAHH and SAH into ECL system.
• In situ generating l-Hcys greatly amplified the ECL signal.
• In situ generated l-Hcys could gradually accumulate on the surface of the immunosensor.
• The strategy has the advantages of sensitivity, good selectivity and reproducibility.
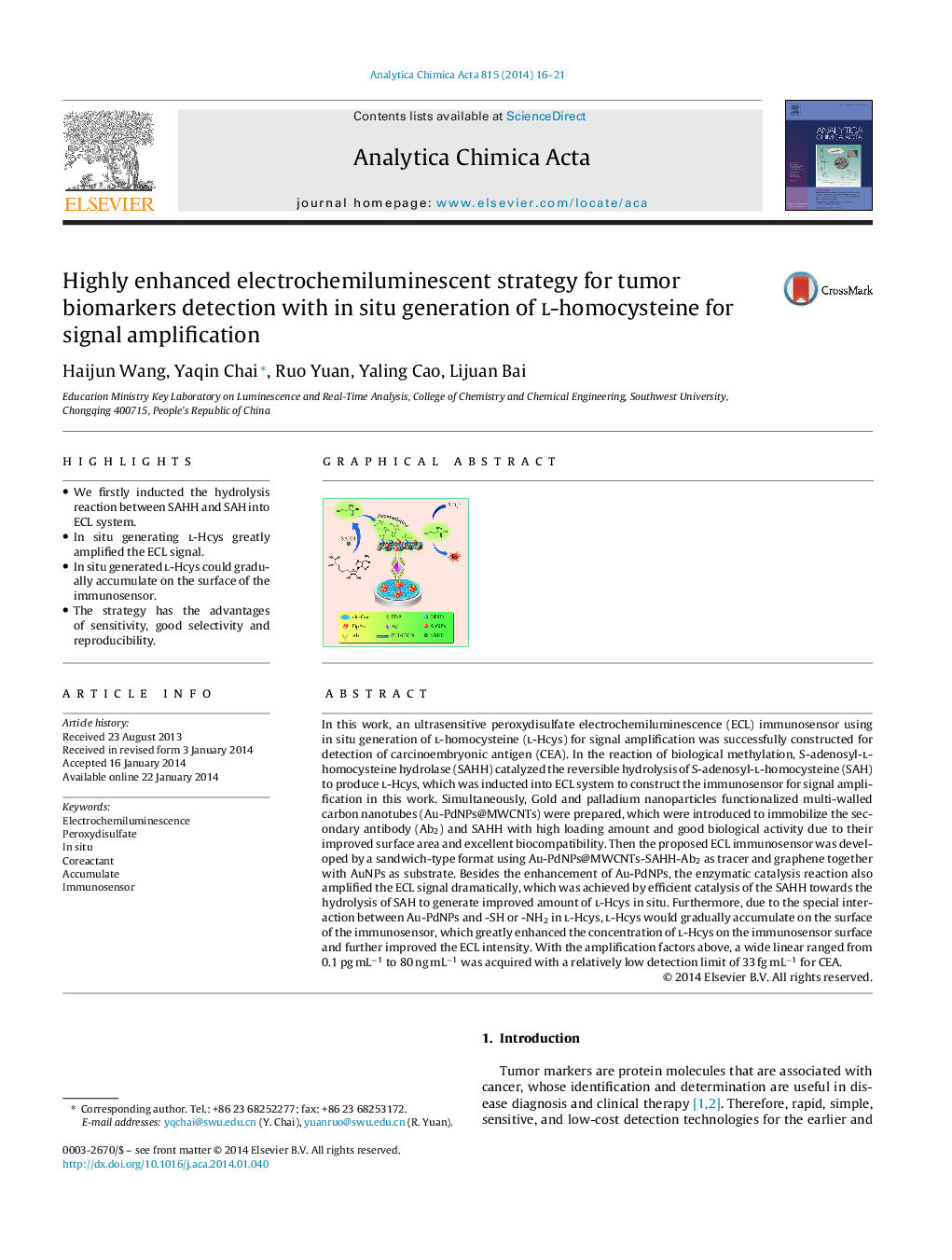
In this work, an ultrasensitive peroxydisulfate electrochemiluminescence (ECL) immunosensor using in situ generation of l-homocysteine (l-Hcys) for signal amplification was successfully constructed for detection of carcinoembryonic antigen (CEA). In the reaction of biological methylation, S-adenosyl-l-homocysteine hydrolase (SAHH) catalyzed the reversible hydrolysis of S-adenosyl-l-homocysteine (SAH) to produce l-Hcys, which was inducted into ECL system to construct the immunosensor for signal amplification in this work. Simultaneously, Gold and palladium nanoparticles functionalized multi-walled carbon nanotubes (Au-PdNPs@MWCNTs) were prepared, which were introduced to immobilize the secondary antibody (Ab2) and SAHH with high loading amount and good biological activity due to their improved surface area and excellent biocompatibility. Then the proposed ECL immunosensor was developed by a sandwich-type format using Au-PdNPs@MWCNTs-SAHH-Ab2 as tracer and graphene together with AuNPs as substrate. Besides the enhancement of Au-PdNPs, the enzymatic catalysis reaction also amplified the ECL signal dramatically, which was achieved by efficient catalysis of the SAHH towards the hydrolysis of SAH to generate improved amount of l-Hcys in situ. Furthermore, due to the special interaction between Au-PdNPs and -SH or -NH2 in l-Hcys, l-Hcys would gradually accumulate on the surface of the immunosensor, which greatly enhanced the concentration of l-Hcys on the immunosensor surface and further improved the ECL intensity. With the amplification factors above, a wide linear ranged from 0.1 pg mL−1 to 80 ng mL−1 was acquired with a relatively low detection limit of 33 fg mL−1 for CEA.
Figure optionsDownload as PowerPoint slide
Journal: Analytica Chimica Acta - Volume 815, 7 March 2014, Pages 16–21