| کد مقاله | کد نشریه | سال انتشار | مقاله انگلیسی | نسخه تمام متن |
|---|---|---|---|---|
| 1402427 | 1501743 | 2014 | 10 صفحه PDF | دانلود رایگان |
• Two new copper(II) complexes have been synthesized.
• Complex 1 is square planar and complex 2 is square pyramidal.
• Interactions of 1 and 2 with CT DNA were studied by UV–VIS spectroscopy.
• 1 interacted better with CT DNA than 2.
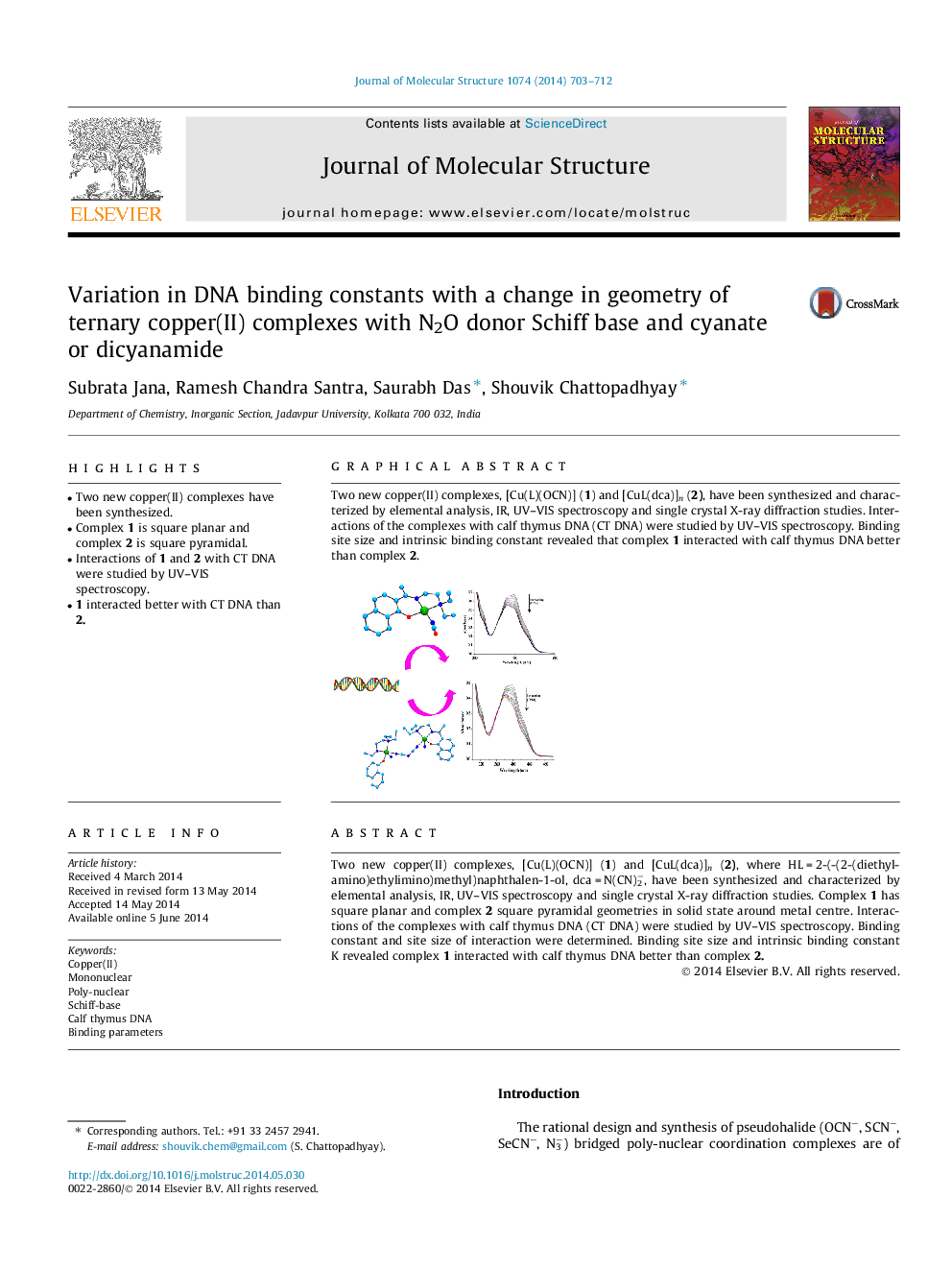
Two new copper(II) complexes, [Cu(L)(OCN)] (1) and [CuL(dca)]n (2), where HL = 2-(-(2-(diethylamino)ethylimino)methyl)naphthalen-1-ol, dca = N(CN)2−, have been synthesized and characterized by elemental analysis, IR, UV–VIS spectroscopy and single crystal X-ray diffraction studies. Complex 1 has square planar and complex 2 square pyramidal geometries in solid state around metal centre. Interactions of the complexes with calf thymus DNA (CT DNA) were studied by UV–VIS spectroscopy. Binding constant and site size of interaction were determined. Binding site size and intrinsic binding constant K revealed complex 1 interacted with calf thymus DNA better than complex 2.
Two new copper(II) complexes, [Cu(L)(OCN)] (1) and [CuL(dca)]n (2), have been synthesized and characterized by elemental analysis, IR, UV–VIS spectroscopy and single crystal X-ray diffraction studies. Interactions of the complexes with calf thymus DNA (CT DNA) were studied by UV–VIS spectroscopy. Binding site size and intrinsic binding constant revealed that complex 1 interacted with calf thymus DNA better than complex 2.Figure optionsDownload as PowerPoint slide
Journal: Journal of Molecular Structure - Volume 1074, 25 September 2014, Pages 703–712