| کد مقاله | کد نشریه | سال انتشار | مقاله انگلیسی | نسخه تمام متن |
|---|---|---|---|---|
| 1402776 | 1501755 | 2014 | 7 صفحه PDF | دانلود رایگان |
• A new succinate bridged 1D coordination polymer of cobalt(II) is isolated.
• X-ray study reveals interesting 2D sheet and 3D network structures.
• Variable-temperature magnetic susceptibility shows weak antiferromagnetic coupling.
• Thermogravimetric analysis proves the thermal stability and decomposition pattern.
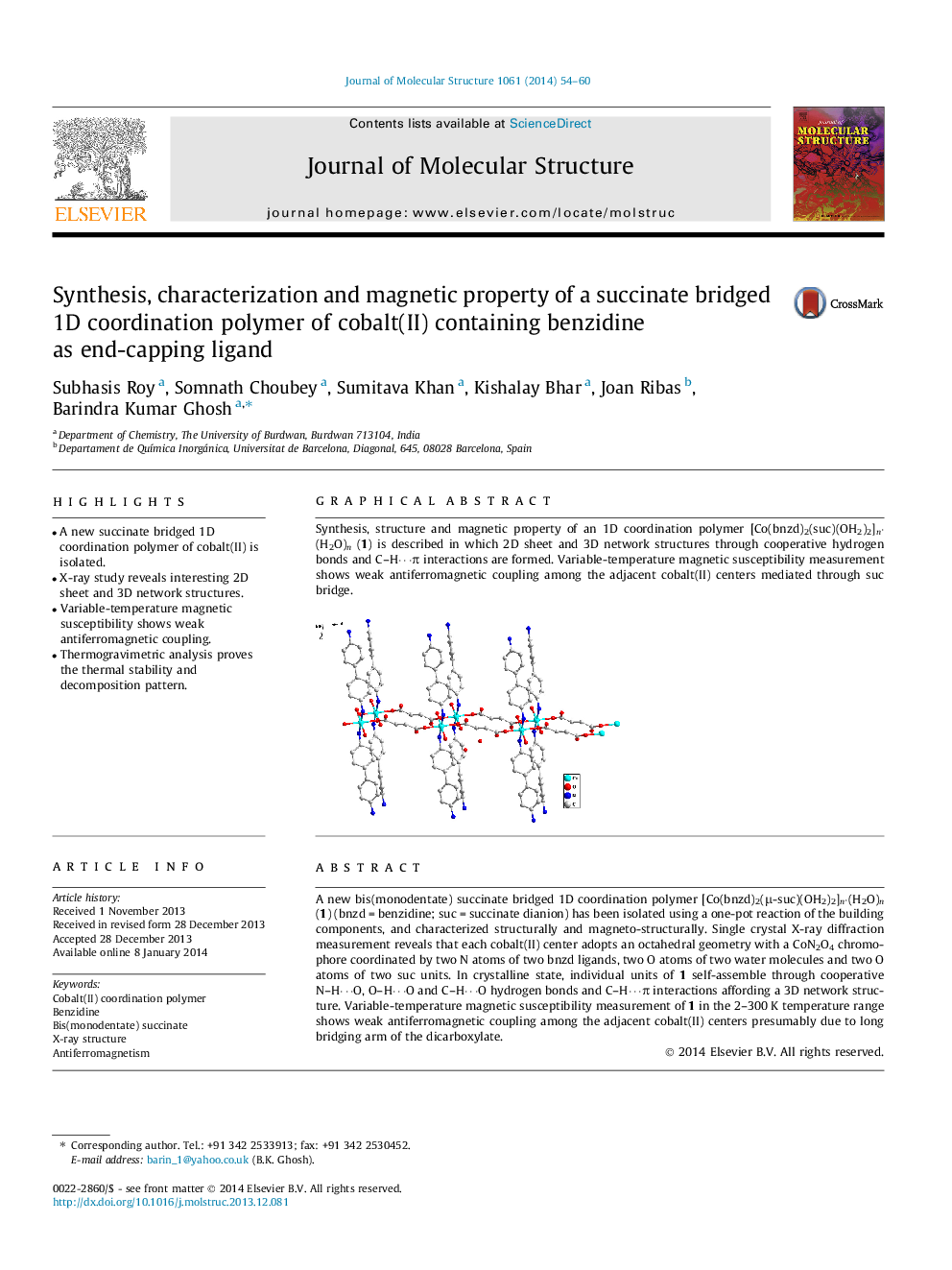
A new bis(monodentate) succinate bridged 1D coordination polymer [Co(bnzd)2(μ-suc)(OH2)2]n⋅(H2O)n (1) (bnzd = benzidine; suc = succinate dianion) has been isolated using a one-pot reaction of the building components, and characterized structurally and magneto-structurally. Single crystal X-ray diffraction measurement reveals that each cobalt(II) center adopts an octahedral geometry with a CoN2O4 chromophore coordinated by two N atoms of two bnzd ligands, two O atoms of two water molecules and two O atoms of two suc units. In crystalline state, individual units of 1 self-assemble through cooperative N–H⋯O, O–H⋯O and C–H⋯O hydrogen bonds and C–H⋯π interactions affording a 3D network structure. Variable-temperature magnetic susceptibility measurement of 1 in the 2–300 K temperature range shows weak antiferromagnetic coupling among the adjacent cobalt(II) centers presumably due to long bridging arm of the dicarboxylate.
Synthesis, structure and magnetic property of an 1D coordination polymer [Co(bnzd)2(suc)(OH2)2]n·(H2O)n (1) is described in which 2D sheet and 3D network structures through cooperative hydrogen bonds and C–H⋯π interactions are formed. Variable-temperature magnetic susceptibility measurement shows weak antiferromagnetic coupling among the adjacent cobalt(II) centers mediated through suc bridge.Figure optionsDownload as PowerPoint slide
Journal: Journal of Molecular Structure - Volume 1061, 5 March 2014, Pages 54–60