| کد مقاله | کد نشریه | سال انتشار | مقاله انگلیسی | نسخه تمام متن |
|---|---|---|---|---|
| 1408369 | 1501736 | 2015 | 7 صفحه PDF | دانلود رایگان |
• The fourth member of the compounds POX3 with X = I was synthesized and characterized by infrared spectroscopy.
• The molecular structure of POX3 (X = F, Cl, Br, I) molecules were investigated by means DFT and ab-initio methods.
• NBO analysis was performed in order to know the hyper-conjugative interactions that favor one structure over another.
• HOMO and LUMO analysis have been performed to know global descriptors and reactivity of the molecules.
The fourth member of the series of compounds of the type POX3 with X = I was synthesized and characterized by infrared spectroscopy. The geometrical parameters and vibrational properties of POX3 (X = F, Cl, Br, I) molecules were investigated theoretically by means DFT and ab initio methods. Available geometrical and vibrational data were used together with theoretical calculations in order to obtain a set of scaled force constants. The observed trends in geometrical parameters are analyzed and compared with those obtained in a previous work for the VOX3 (X = F, Cl, Br, I) series of compounds. NBO analysis was performed in order to know the hyper-conjugative interactions that favor one structure over another. The molecular properties such as ionization potential, electron affinity, electronegativity, chemical potential, chemical hardness, softness and global electrophilicity index have been deduced from HOMO–LUMO analysis.
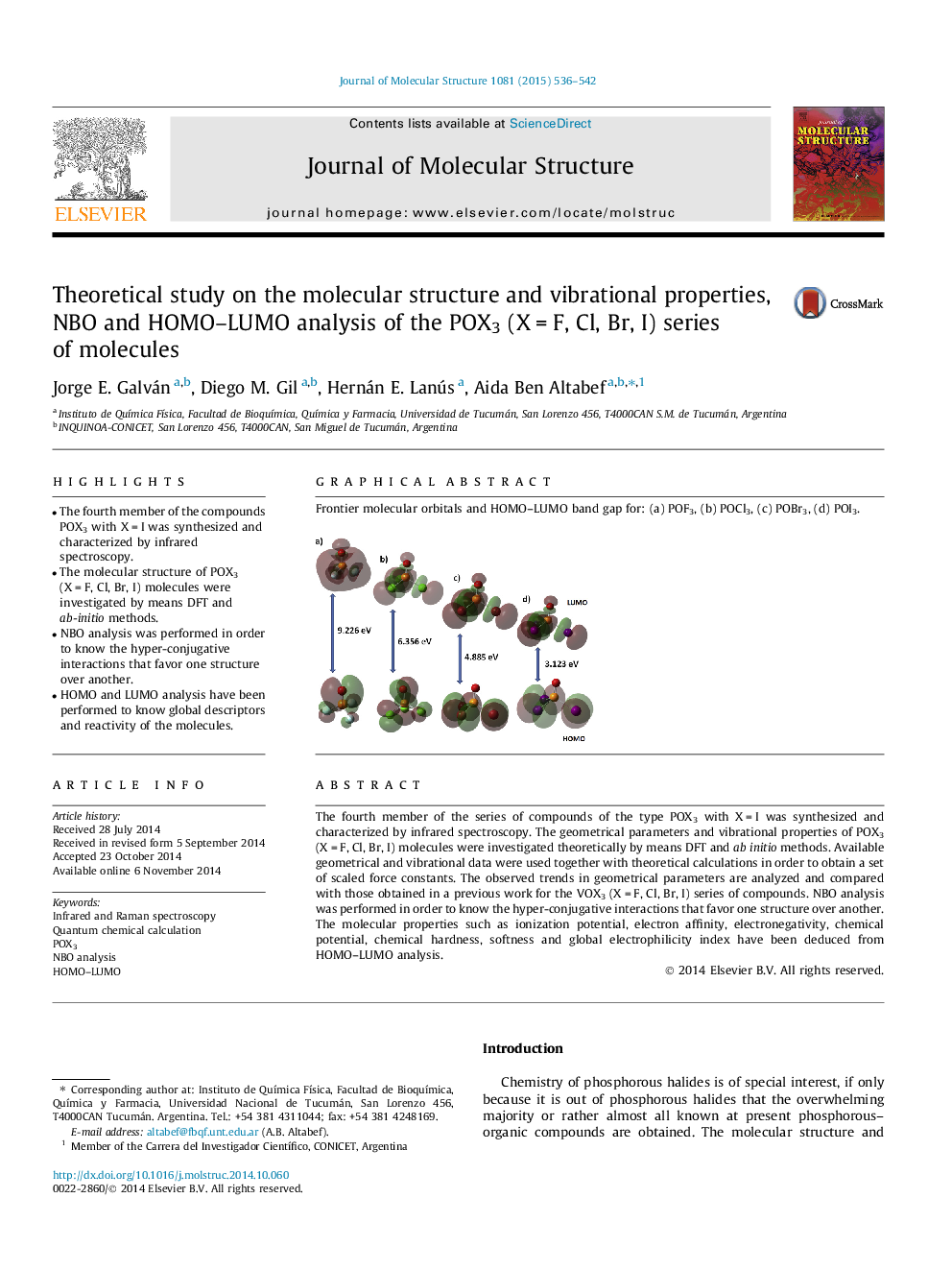
Frontier molecular orbitals and HOMO–LUMO band gap for: (a) POF3, (b) POCl3, (c) POBr3, (d) POI3.Figure optionsDownload as PowerPoint slide
Journal: Journal of Molecular Structure - Volume 1081, 5 February 2015, Pages 536–542