| کد مقاله | کد نشریه | سال انتشار | مقاله انگلیسی | نسخه تمام متن |
|---|---|---|---|---|
| 5223999 | 1383504 | 2010 | 9 صفحه PDF | دانلود رایگان |
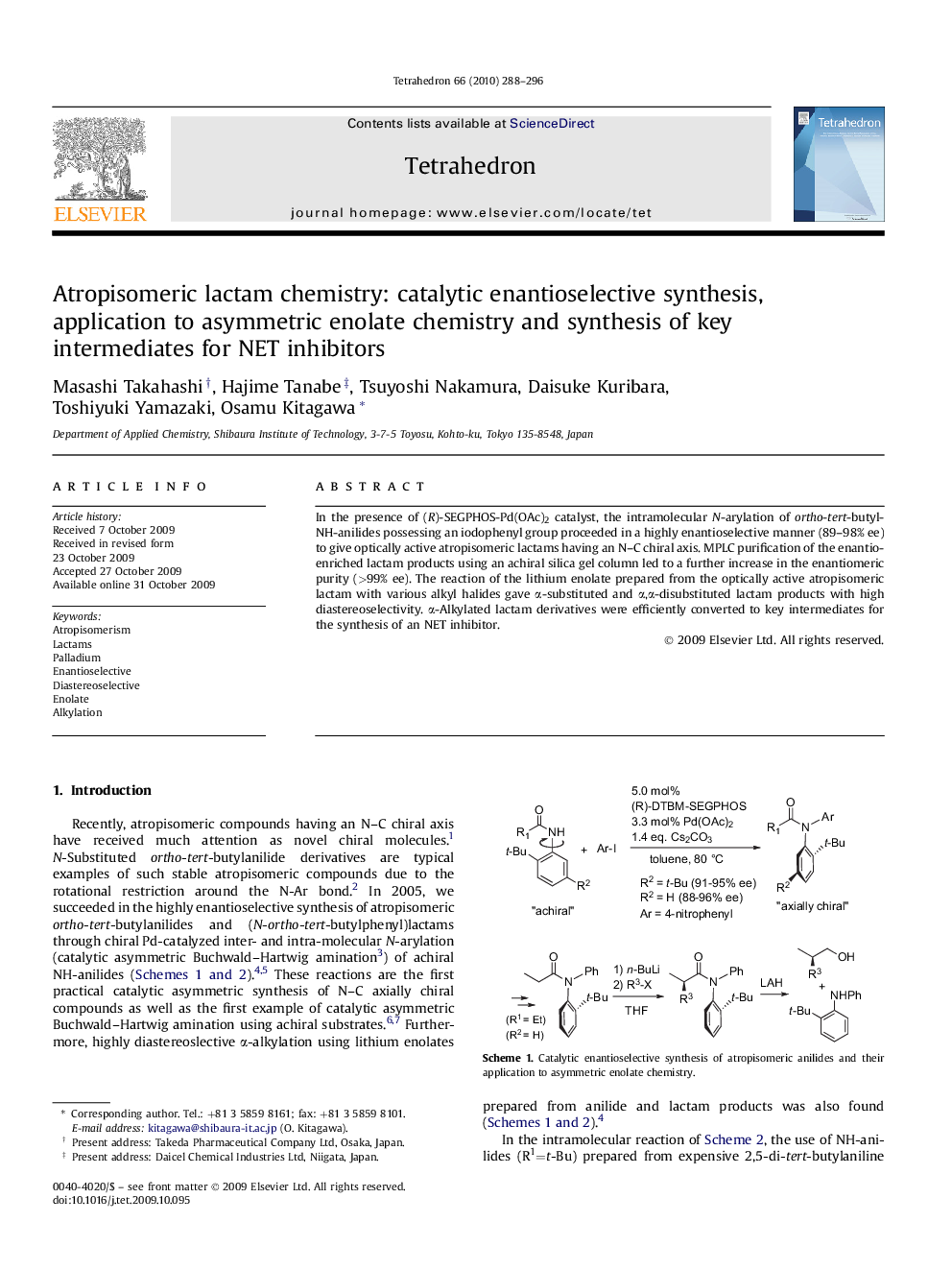
In the presence of (R)-SEGPHOS-Pd(OAc)2 catalyst, the intramolecular N-arylation of ortho-tert-butyl-NH-anilides possessing an iodophenyl group proceeded in a highly enantioselective manner (89-98% ee) to give optically active atropisomeric lactams having an N-C chiral axis. MPLC purification of the enantio-enriched lactam products using an achiral silica gel column led to a further increase in the enantiomeric purity (>99% ee). The reaction of the lithium enolate prepared from the optically active atropisomeric lactam with various alkyl halides gave α-substituted and α,α-disubstituted lactam products with high diastereoselectivity. α-Alkylated lactam derivatives were efficiently converted to key intermediates for the synthesis of an NET inhibitor.
Journal: Tetrahedron - Volume 66, Issue 1, 2 January 2010, Pages 288-296