| کد مقاله | کد نشریه | سال انتشار | مقاله انگلیسی | نسخه تمام متن |
|---|---|---|---|---|
| 5271491 | 1385440 | 2013 | 7 صفحه PDF | دانلود رایگان |
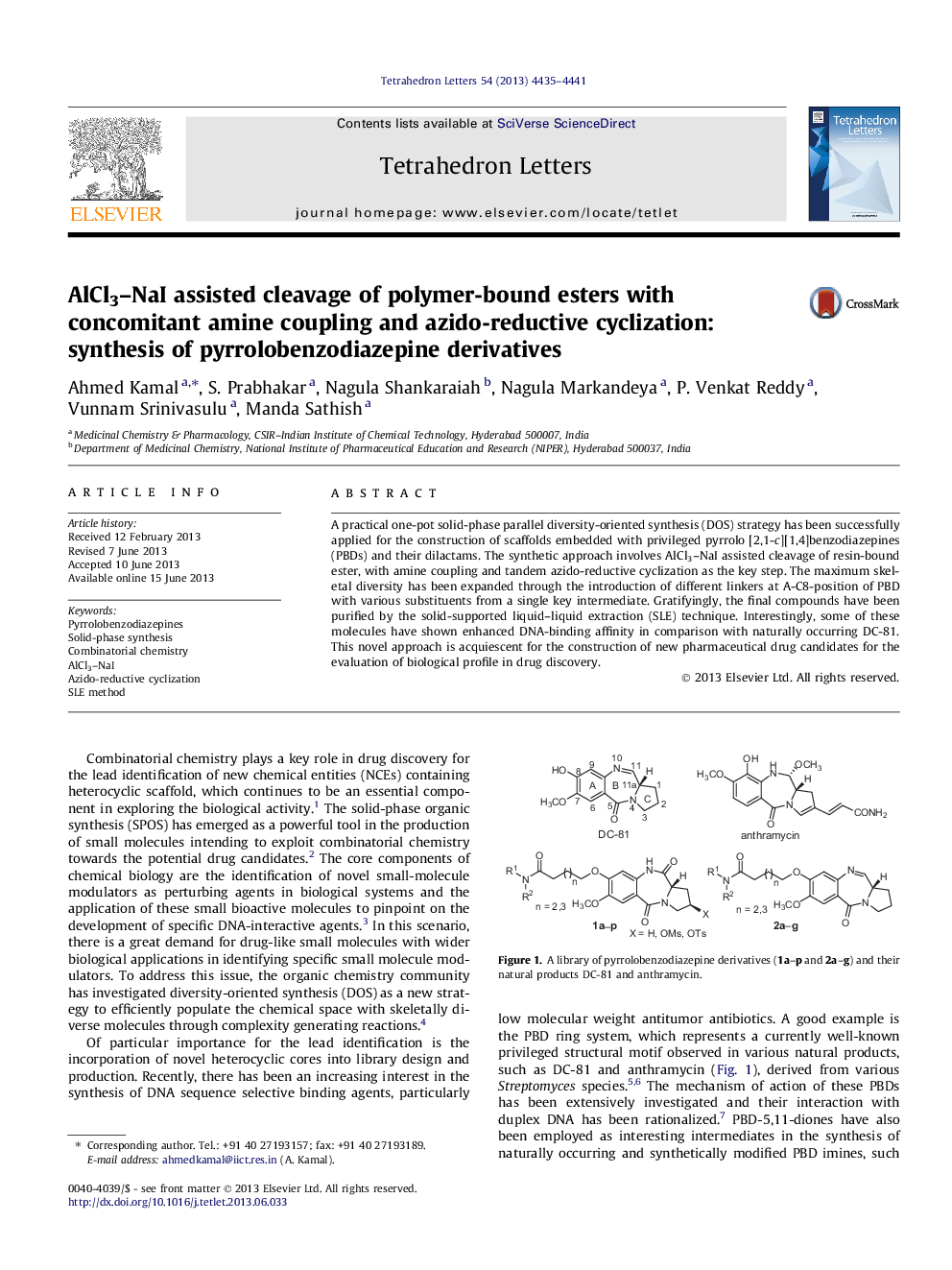
A practical one-pot solid-phase parallel diversity-oriented synthesis (DOS) strategy has been successfully applied for the construction of scaffolds embedded with privileged pyrrolo [2,1-c][1,4]benzodiazepines (PBDs) and their dilactams. The synthetic approach involves AlCl3-NaI assisted cleavage of resin-bound ester, with amine coupling and tandem azido-reductive cyclization as the key step. The maximum skeletal diversity has been expanded through the introduction of different linkers at A-C8-position of PBD with various substituents from a single key intermediate. Gratifyingly, the final compounds have been purified by the solid-supported liquid-liquid extraction (SLE) technique. Interestingly, some of these molecules have shown enhanced DNA-binding affinity in comparison with naturally occurring DC-81. This novel approach is acquiescent for the construction of new pharmaceutical drug candidates for the evaluation of biological profile in drug discovery.
Journal: Tetrahedron Letters - Volume 54, Issue 33, 14 August 2013, Pages 4435-4441