| کد مقاله | کد نشریه | سال انتشار | مقاله انگلیسی | نسخه تمام متن |
|---|---|---|---|---|
| 591809 | 1453883 | 2016 | 10 صفحه PDF | دانلود رایگان |
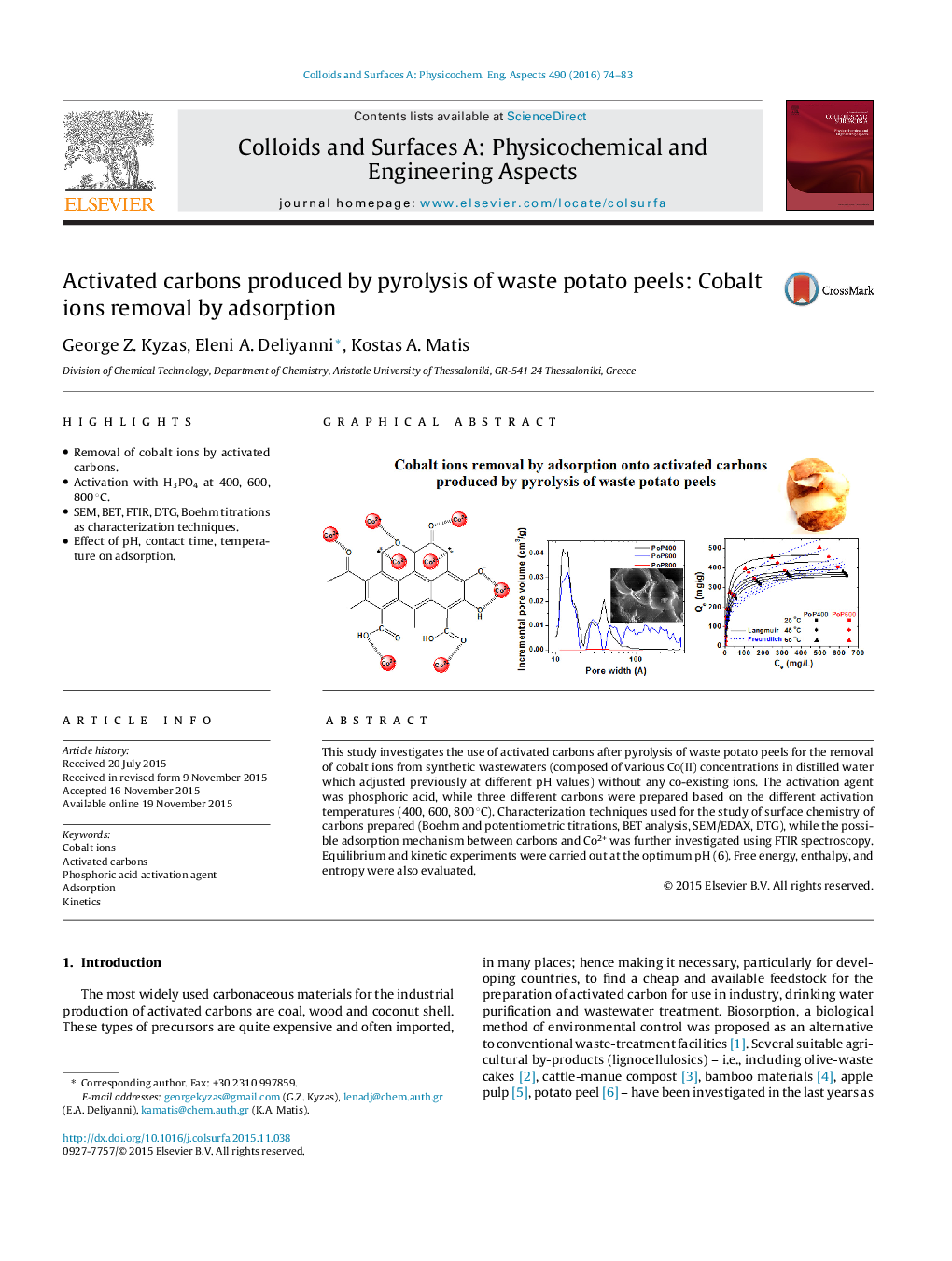
• Removal of cobalt ions by activated carbons.
• Activation with H3PO4 at 400, 600, 800 °C.
• SEM, BET, FTIR, DTG, Boehm titrations as characterization techniques.
• Effect of pH, contact time, temperature on adsorption.
This study investigates the use of activated carbons after pyrolysis of waste potato peels for the removal of cobalt ions from synthetic wastewaters (composed of various Co(II) concentrations in distilled water which adjusted previously at different pH values) without any co-existing ions. The activation agent was phosphoric acid, while three different carbons were prepared based on the different activation temperatures (400, 600, 800 °C). Characterization techniques used for the study of surface chemistry of carbons prepared (Boehm and potentiometric titrations, BET analysis, SEM/EDAX, DTG), while the possible adsorption mechanism between carbons and Co2+ was further investigated using FTIR spectroscopy. Equilibrium and kinetic experiments were carried out at the optimum pH (6). Free energy, enthalpy, and entropy were also evaluated.
Figure optionsDownload as PowerPoint slide
Journal: Colloids and Surfaces A: Physicochemical and Engineering Aspects - Volume 490, 5 February 2016, Pages 74–83