| کد مقاله | کد نشریه | سال انتشار | مقاله انگلیسی | نسخه تمام متن |
|---|---|---|---|---|
| 592074 | 1453891 | 2015 | 5 صفحه PDF | دانلود رایگان |
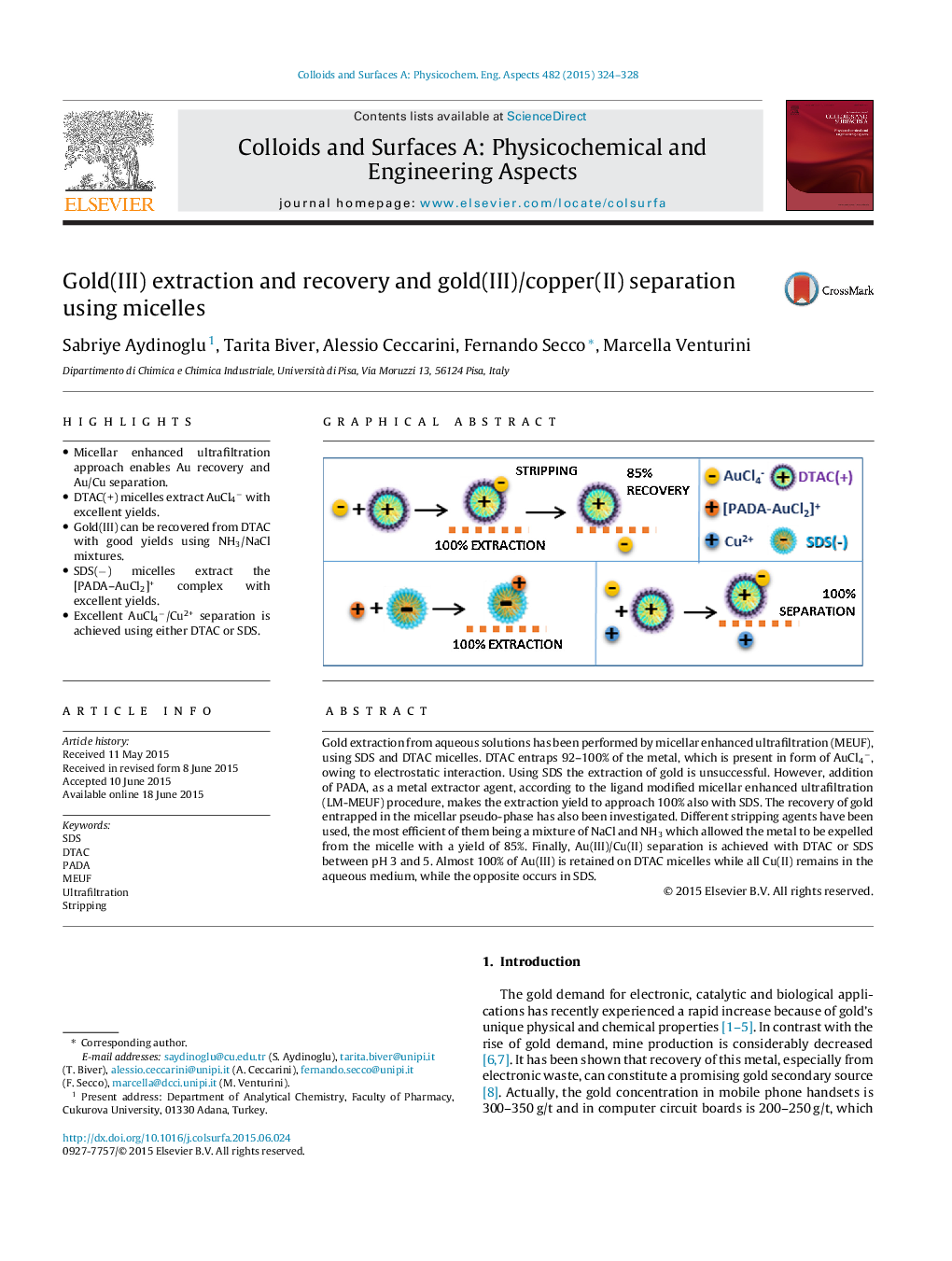
• Micellar enhanced ultrafiltration approach enables Au recovery and Au/Cu separation.
• DTAC(+) micelles extract AuCl4− with excellent yields.
• Gold(III) can be recovered from DTAC with good yields using NH3/NaCl mixtures.
• SDS(−) micelles extract the [PADA–AuCl2]+ complex with excellent yields.
• Excellent AuCl4−/Cu2+ separation is achieved using either DTAC or SDS.
Gold extraction from aqueous solutions has been performed by micellar enhanced ultrafiltration (MEUF), using SDS and DTAC micelles. DTAC entraps 92–100% of the metal, which is present in form of AuCl4−, owing to electrostatic interaction. Using SDS the extraction of gold is unsuccessful. However, addition of PADA, as a metal extractor agent, according to the ligand modified micellar enhanced ultrafiltration (LM-MEUF) procedure, makes the extraction yield to approach 100% also with SDS. The recovery of gold entrapped in the micellar pseudo-phase has also been investigated. Different stripping agents have been used, the most efficient of them being a mixture of NaCl and NH3 which allowed the metal to be expelled from the micelle with a yield of 85%. Finally, Au(III)/Cu(II) separation is achieved with DTAC or SDS between pH 3 and 5. Almost 100% of Au(III) is retained on DTAC micelles while all Cu(II) remains in the aqueous medium, while the opposite occurs in SDS.
Figure optionsDownload as PowerPoint slide
Journal: Colloids and Surfaces A: Physicochemical and Engineering Aspects - Volume 482, 5 October 2015, Pages 324–328