| کد مقاله | کد نشریه | سال انتشار | مقاله انگلیسی | نسخه تمام متن |
|---|---|---|---|---|
| 592301 | 1453899 | 2015 | 9 صفحه PDF | دانلود رایگان |
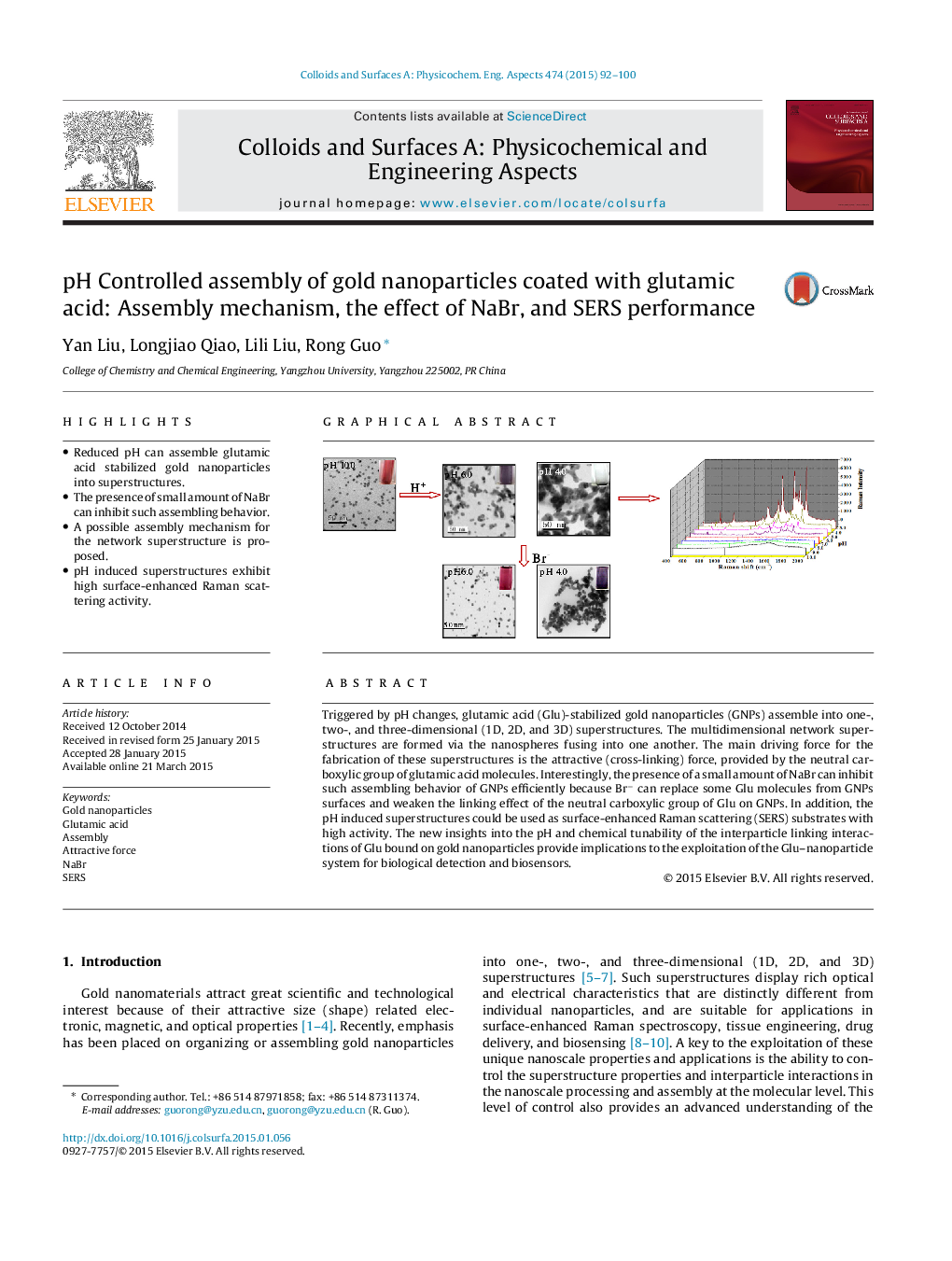
• Reduced pH can assemble glutamic acid stabilized gold nanoparticles into superstructures.
• The presence of small amount of NaBr can inhibit such assembling behavior.
• A possible assembly mechanism for the network superstructure is proposed.
• pH induced superstructures exhibit high surface-enhanced Raman scattering activity.
Triggered by pH changes, glutamic acid (Glu)-stabilized gold nanoparticles (GNPs) assemble into one-, two-, and three-dimensional (1D, 2D, and 3D) superstructures. The multidimensional network superstructures are formed via the nanospheres fusing into one another. The main driving force for the fabrication of these superstructures is the attractive (cross-linking) force, provided by the neutral carboxylic group of glutamic acid molecules. Interestingly, the presence of a small amount of NaBr can inhibit such assembling behavior of GNPs efficiently because Br− can replace some Glu molecules from GNPs surfaces and weaken the linking effect of the neutral carboxylic group of Glu on GNPs. In addition, the pH induced superstructures could be used as surface-enhanced Raman scattering (SERS) substrates with high activity. The new insights into the pH and chemical tunability of the interparticle linking interactions of Glu bound on gold nanoparticles provide implications to the exploitation of the Glu–nanoparticle system for biological detection and biosensors.
Figure optionsDownload as PowerPoint slide
Journal: Colloids and Surfaces A: Physicochemical and Engineering Aspects - Volume 474, 5 June 2015, Pages 92–100