| کد مقاله | کد نشریه | سال انتشار | مقاله انگلیسی | نسخه تمام متن |
|---|---|---|---|---|
| 592285 | 1453898 | 2015 | 6 صفحه PDF | دانلود رایگان |
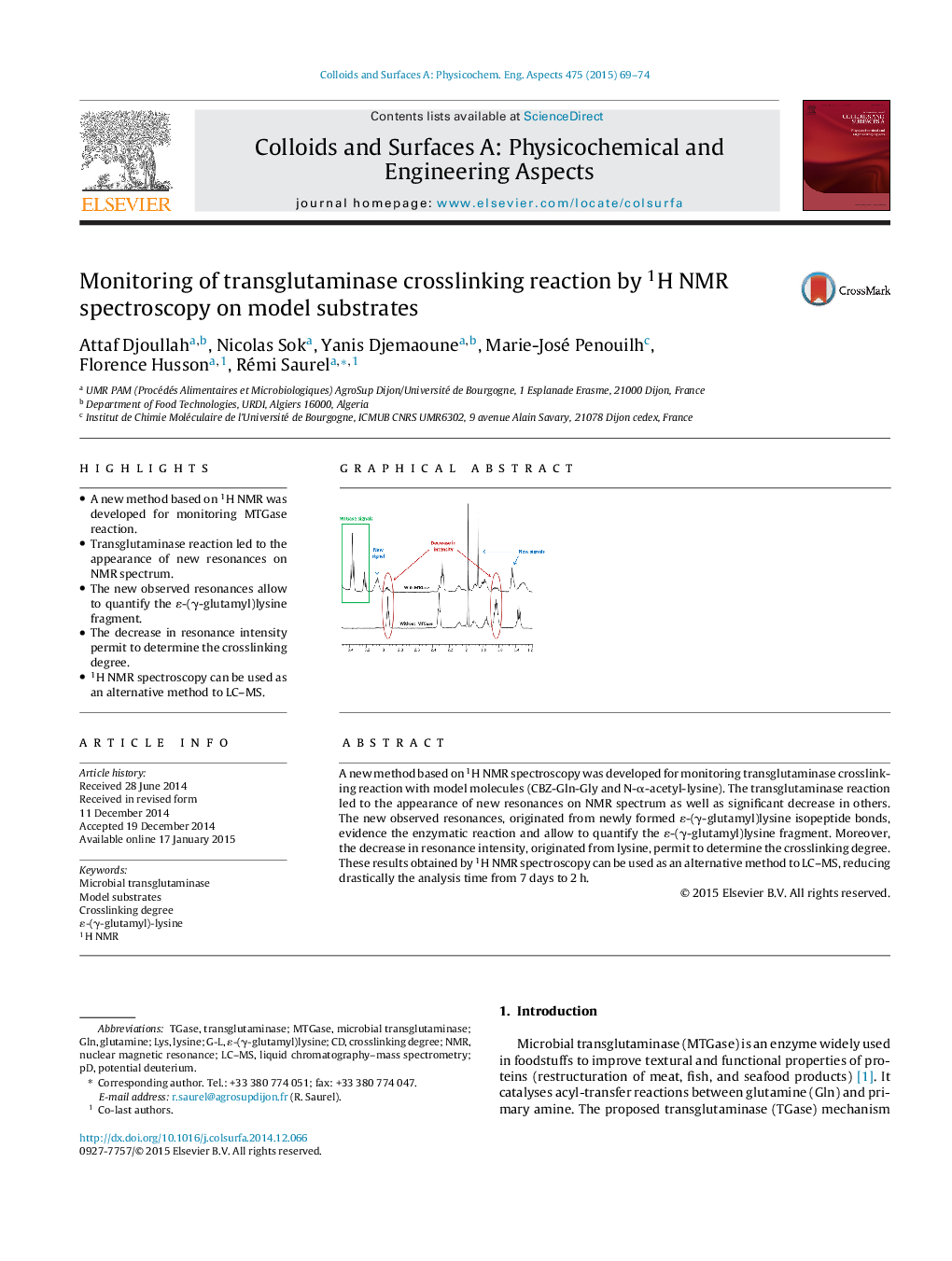
• A new method based on 1H NMR was developed for monitoring MTGase reaction.
• Transglutaminase reaction led to the appearance of new resonances on NMR spectrum.
• The new observed resonances allow to quantify the ɛ-(γ-glutamyl)lysine fragment.
• The decrease in resonance intensity permit to determine the crosslinking degree.
• 1H NMR spectroscopy can be used as an alternative method to LC–MS.
A new method based on 1H NMR spectroscopy was developed for monitoring transglutaminase crosslinking reaction with model molecules (CBZ-Gln-Gly and N-α-acetyl-lysine). The transglutaminase reaction led to the appearance of new resonances on NMR spectrum as well as significant decrease in others. The new observed resonances, originated from newly formed ɛ-(γ-glutamyl)lysine isopeptide bonds, evidence the enzymatic reaction and allow to quantify the ɛ-(γ-glutamyl)lysine fragment. Moreover, the decrease in resonance intensity, originated from lysine, permit to determine the crosslinking degree. These results obtained by 1H NMR spectroscopy can be used as an alternative method to LC–MS, reducing drastically the analysis time from 7 days to 2 h.
Figure optionsDownload as PowerPoint slide
Journal: Colloids and Surfaces A: Physicochemical and Engineering Aspects - Volume 475, 20 June 2015, Pages 69–74