| کد مقاله | کد نشریه | سال انتشار | مقاله انگلیسی | نسخه تمام متن |
|---|---|---|---|---|
| 69389 | 48761 | 2015 | 6 صفحه PDF | دانلود رایگان |
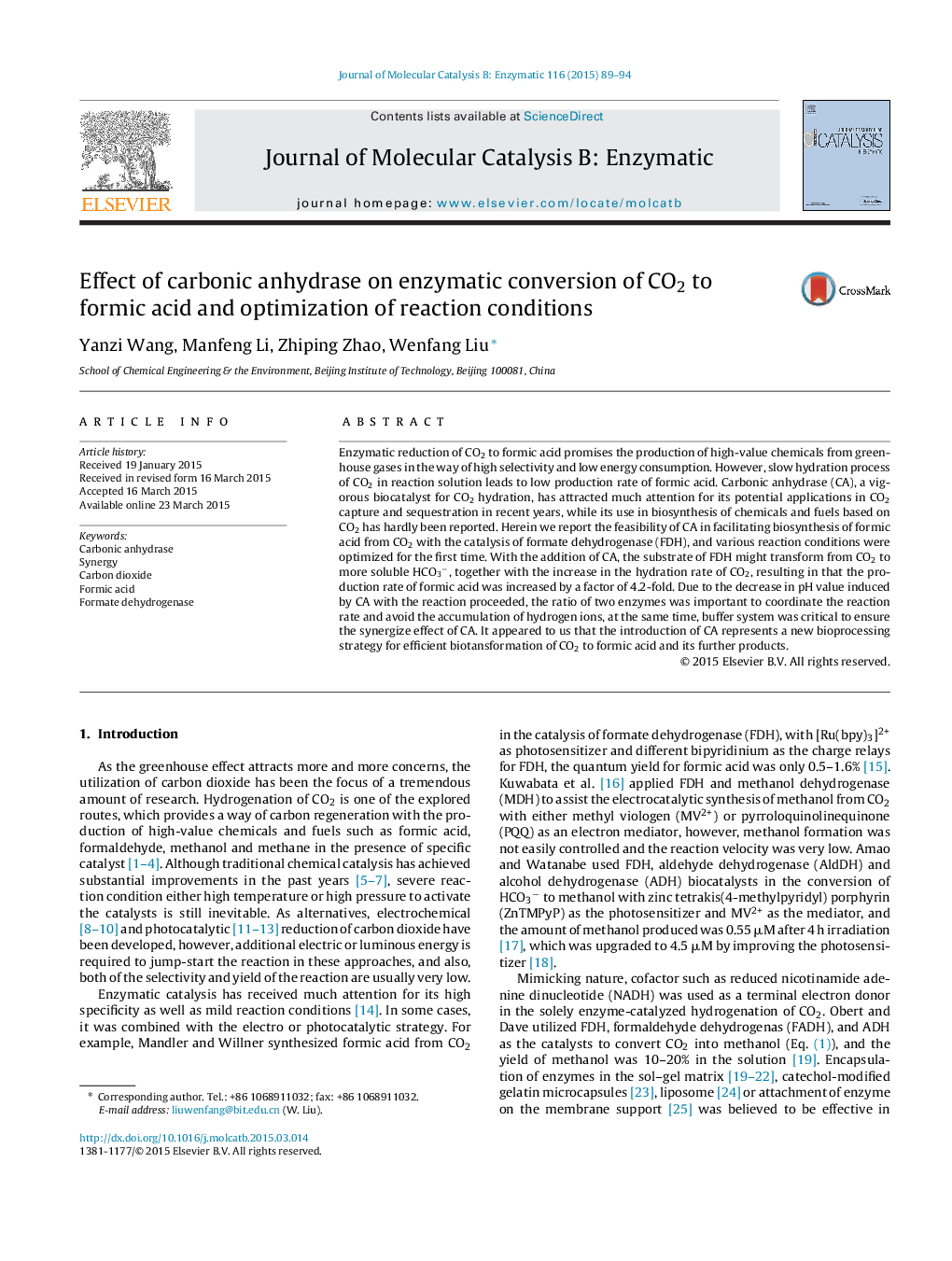
• CA facilitated the enzymatic conversion of CO2 into formic acid, the reaction rate was increased by a factor of 4.2-fold.
• The reaction conditions were optimized.
• A higher concentration of CA caused the decrease in the overall reaction rate, increasing FDH/CA was favorable to the reaction.
• PB was better than the other several systems, and pH value and the concentration needed be controlled strictly.
• The maximum rate was achieved at 37 °C in 0.05 M PB with pH value of 7.0.
Enzymatic reduction of CO2 to formic acid promises the production of high-value chemicals from greenhouse gases in the way of high selectivity and low energy consumption. However, slow hydration process of CO2 in reaction solution leads to low production rate of formic acid. Carbonic anhydrase (CA), a vigorous biocatalyst for CO2 hydration, has attracted much attention for its potential applications in CO2 capture and sequestration in recent years, while its use in biosynthesis of chemicals and fuels based on CO2 has hardly been reported. Herein we report the feasibility of CA in facilitating biosynthesis of formic acid from CO2 with the catalysis of formate dehydrogenase (FDH), and various reaction conditions were optimized for the first time. With the addition of CA, the substrate of FDH might transform from CO2 to more soluble HCO3−, together with the increase in the hydration rate of CO2, resulting in that the production rate of formic acid was increased by a factor of 4.2-fold. Due to the decrease in pH value induced by CA with the reaction proceeded, the ratio of two enzymes was important to coordinate the reaction rate and avoid the accumulation of hydrogen ions, at the same time, buffer system was critical to ensure the synergize effect of CA. It appeared to us that the introduction of CA represents a new bioprocessing strategy for efficient biotansformation of CO2 to formic acid and its further products.
Figure optionsDownload as PowerPoint slide
Journal: Journal of Molecular Catalysis B: Enzymatic - Volume 116, June 2015, Pages 89–94